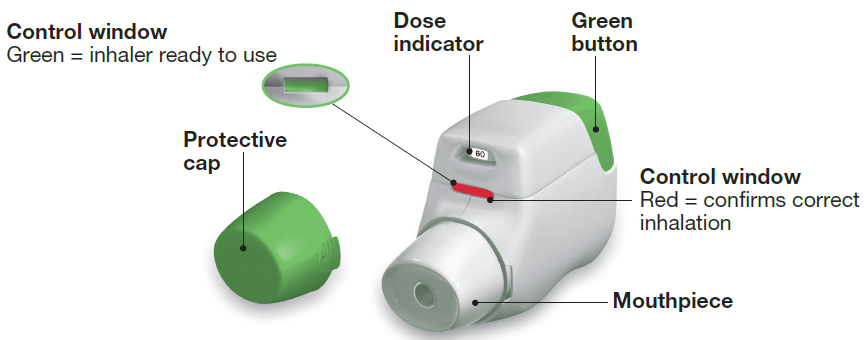
Figure 2 from Aclidinium bromide inhalation powder (tudorza): a long-acting anticholinergic for the management of chronic obstructive pulmonary disease. | Semantic Scholar

US Food and Drug Administration Approves DUAKLIR® PRESSAIR® (aclidinium/formoterol) for Patients with Chronic Obstructive Pulmonary Disease (COPD) | Business Wire

Lung Deposition of Aclidinium Bromide from Genuair®, a Multidose Dry Powder Inhaler | Semantic Scholar
FDA Approves TUDORZA® PRESSAIR® (aclidinium bromide inhalation powder) Supplemental New Drug Application (sNDA) to include data for the Reduction of COPD Exacerbations and Hospitalizations | BioSpace

Evaluation of the Features of the Aclidinium Bromide Inhaler According... | Download Scientific Diagram

Aclidinium Bromide and Formoterol Fumarate Approved for COPD Maintenance Treatment - Pulmonology Advisor